Covid Booster Dose Trial
19Covid booster trial will give third vaccine dose to UK volunteers Scientists to test seven vaccines for possible protection against new variants in effort to avert winter surge in infections A. At a dose of 30µg as studied in the Phase 23 study C4591001 In healthy adults 16 years of age and older.
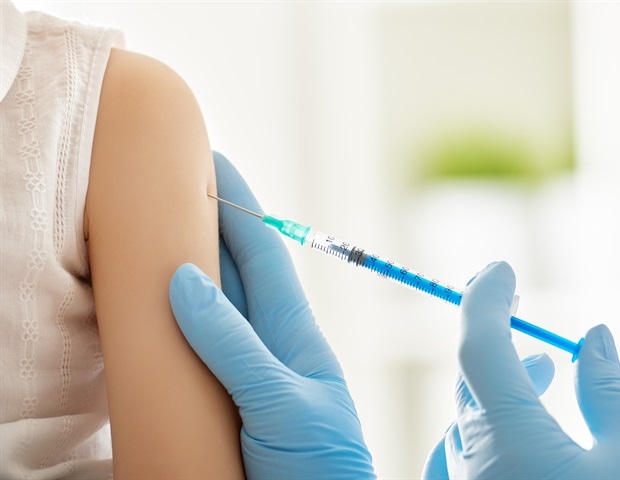
World First Trial Of Third Dose Covid 19 Booster Vaccine Shows Improved Protection In Transplant Patients
1The National Institutes of Health has started a Phase 12 clinical trial in which adult volunteers who have been fully vaccinated against COVID-19 will receive booster doses of different COVID-19 vaccines to determine the safety and immunogenicity of mixed boosted regimens.

Covid booster dose trial. By Mansur Shaheen For DailymailCom. Third dose also increased antibody levels five-fold in 18-to-55-year-olds and 11-fold for those aged 65 to 85. 20The COV-Boost trial is being run by the University of Southampton and theyre taking volunteers from people across the country via their website.
16Participants involved in the trial received a booster dose 8 to 9 months after receiving their second dose of vaccine. 19The trial will look at seven different COVID-19 vaccines as potential boosters given at least 10 to 12 weeks after a second dose as part of the ongoing vaccination programme. 15Scientists with the Ajmera Transplant Centre at UHN have conducted a first-in-the-world randomized placebo-controlled trial of third dose COVID-19 booster vaccine.
The duration of the study for each participant will be up to. Participants involved in the trial received a booster dose 8 to 9 months after receiving their second dose of vaccine. 8They will receive the EUA Moderna vaccine followed by a booster vaccine from Moderna or another vaccine manufacturer after at least 12 weeks.
16Pfizer submits initial trial data to FDA for COVID-19 vaccine booster that showed third dose increased antibody levels against Indian Delta variant compared to two doses. 9The study is designed to describe vaccine efficacy of a booster dose of BNT162b2 over time against COVID-19. 17Pfizer and BioNTech have submitted preliminary clinical trial data for their combined COVID-19 vaccine booster shot to the US Food and Drug Administration FDA.
Likely to recommend booster shots eight months after second dose Pfizer and BioNTech said Monday they have submitted early stage clinical trial data to the Food and Drug Administration as part. India to evaluate impact of booster dose and jab mix trial Doctors at Christian Medical College will examine the immune responses from a third dose in vaccinated people and a mixed dose schedule in unvaccinated people. 19Third doses of covid-19 vaccines will be rolled out to combat delta variant surge in US.
16Pfizer submits initial trial data to FDA for COVID-19 vaccine booster that showed third dose increased antibody levels against Indian Delta variant compared to two doses. 12Adults Needed for UW COVID-19 Vaccine Booster Clinical Trial The University of Washington is seeking volunteers for a Phase 12 COVID-19 vaccine booster trial. Booster vaccinations are scheduled to begin next month.
The data suggests that booster shots elicit higher antibody responses and may be particularly effective against the highly permeable Indian delta variant and the South African beta variant. The US will start making booster vaccines available on 20 September health officials have announced. The purpose of this clinical trial is to test the safety and effectiveness of additional doses of EUA vaccines or boosts in people who have received an EUA vaccine series.
9Pfizer-BioNTech to begin trials of Covid-19 booster vaccine in August 09 Jul 2021 Last Updated July 9th 2021 1148 The booster dose stimulated neutralisation titres five to ten-fold greater than initial doses against the wild type and Beta variants. 16The FDA had already expanded emergency use of Pfizer-BioNTech and Modernas COVID-19 vaccines to authorize a booster shot for this population. 28PITTSBURGH The University of Pittsburgh School of Medicine through the Pittsburgh Vaccine Trials Unit PVTU is one of twelve sites across the country selected to participate in a National Institute of Allergy and Infectious Diseases clinical trial in which fully vaccinated adults will receive a third booster dose of a COVID-19 vaccine.
Booster trials are taking place across the. Individuals interested in enrolling in the trial can call 713-798-4912 or email Covid-vaxbcmedu. 11By Nicole Wetsman Aug 11 2021 1235pm EDT The National Institutes of Health is giving a booster dose to 200 kidney transplant patients who did.
According to the news release the booster dose remained effective against. 20UK launches booster dose trial of seven Covid-19 vaccines 20 May 2021 Last Updated May 20th 2021 1049 The UK has initiated a new trial to assess a third booster dose of seven different Covid-19 vaccines.

Unvaccinated Volunteers Needed For Covid 19 Booster Trial Kpwhri

Trial To Test If Booster Shots Keep Covid 19 At Bay Newsroom

Covid 19 Recommendation Of 3rd Booster Dose Based More On Conjecture Than Data Say Experts
Posting Komentar untuk "Covid Booster Dose Trial"